Evaporative cooling: Energy-efficient removal of excess heat
This is why sweating cools your skin, and this is why a high-pressure air humidifying system has the ability to lower the temperature in a room. This effect – the evaporative cooling effect – is both highly energy-efficient and a strong choice whenever you need to bring down excess heat. This is how you can use the evaporative effect to create a more productive environment in a production facility.
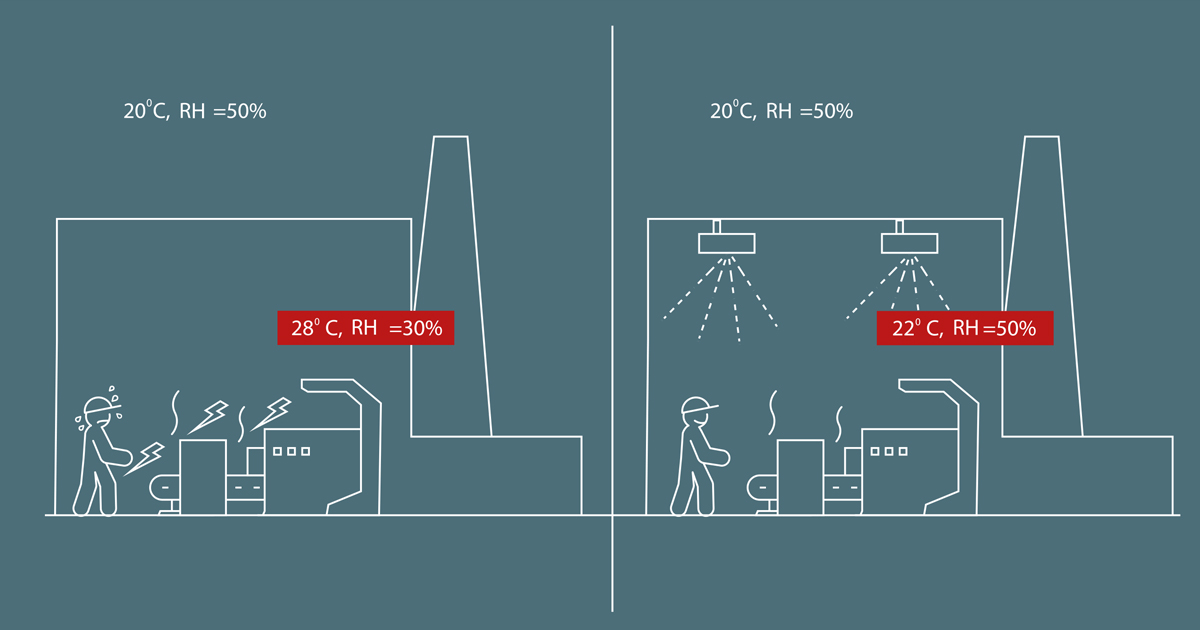
In industrial facilities, many processes generate excess heat. Not only does this drive the temperature up, it also causes relative air humidity to drop with all the negative side-effects this can have: Risk of electrostatic discharge, desiccation of products, reduced comfort for the employees and less control of production processes.
By evaporating water using an air humidifying system, these side-effects can be reduced or eliminated – and the evaporative effect of introducing water spray into a hot room will provide an energy-efficient cooling effect, as well.
How to turn 2 W of electrical energy into 630 W of cooling effect
An air humidifying system using cold water can create a significant cooling effect using very little energy. It is the evaporation of the water that causes the whole system to dissipate energy through an adiabatic process described below – the science behind all this is highly complex, but the effect is both measurable and predictable.
The cooling effect of evaporating water vapor is approx. 630 W of cooling effect per kilogram of water evaporated – and for each kilogram of water, an energy input of approx. 2 W is required in an Airtec® high-pressure air humidifying system. In other words, by exploiting a naturally occurring process, evaporative cooling is a highly energy-efficient method for cooling rooms and production facilities with local heat sources.
This energy efficiency comes from the fact that, unlike conventional air conditioning systems, evaporative cooling happens naturally without the need for compressors and other power-consuming components.
The basics of the adiabatic effectThe adiabatic effect is a result of the first law of thermodynamics, which states that energy in a closed system does not go away, it only goes somewhere. Put another way, in the adiabatic process, energy is transferred through work rather than heat. A compressed gas, for instance, will become hot, and an expanding gas will become cooler.
In the case of a production facility (as shown in the illustration), the presence of local heat sources add heat to the room. In other words, the internal energy of the system increases. To remove it, you can either inject cool air - or you can force the heat to dissipate its energy using the adiabatic effect, which will cause the temperature to fall. If you want to delve into the basics of an adiabatic process, Wikipedia has a splendid scientific definition here >>. |
Air humidifiers can carry a cooling effect in two distinctly different ways – by evaporation or by adding spray to the air flow. Both cool the air by the adiabatic effect we describe above - with the evaporation delivering 630 W of cooling effect per kg of water. The cooling effect of droplets on the floor or in the air, however, is negligible.
Humidifiers are typically installed in a duct or an AHU (air handling unit) or in in-room systems, where vaporizers or atomizers deliver water vapor locally.
Overall, in-room systems are more efficient at exploiting the adiabatic effect and will be more efficient in removing excess heat in a production environment.
See how we’ve done it for others
Airtec® has decades of experience in planning, installing and servicing air humidifying systems – take a look at our case stories here.